OXCT1 regulates NF-κB signaling pathway through β-hydroxybutyrate-mediated ketone body homeostasis in lung cancer


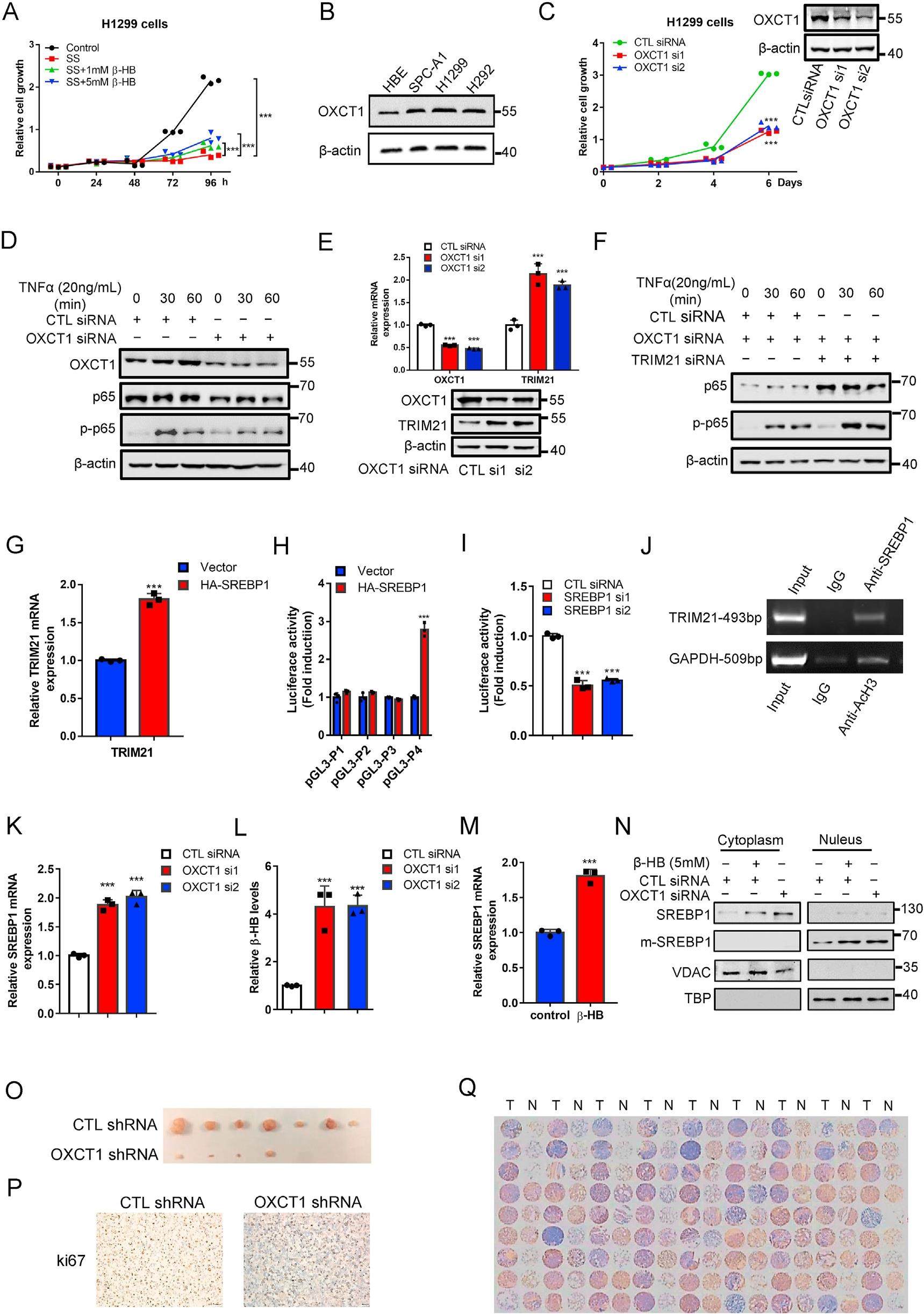
Metabolic reprogramming is one of the hallmarks of cancer. Ketone bodies behave as alternative fuel for cancer cells to support survival and proliferation. 3-Oxoacid CoA-transferase 1 (OXCT1) is a key enzyme that catalyzes the first and rate-limiting step of ketolysis. Recently, several studies have revealed the significance of OXCT1 in cancer development, though the underlying mechanisms remain largely unknown. In this study, we revealed a novel regulatory mechanism for tumorigenesis that OXCT1 regulated SREBP1-TRIM21-p65 axis through ketone body homeostasis in non-small cell lung cancer (NSCLC). In terms of mechanism, we found that OXCT1 could activate NF-κB signaling pathway by suppressing transcriptional activity of the sterol regulatory element binding protein 1 (SREBP1). As a transcription factor, SREBP1 could bind to the promoter of E3 ubiquitin ligase TRIM21, which mediated the ubiquitination of p65. Furthermore, we demonstrated that OXCT1 could maintain the homeostasis of β-hydroxybutyrate (β-HB), which acted as a signaling metabolite to activate SREBP1. Thus, β-HB connected OXCT1 with SREBP1 to activate NF-κB signaling pathway and promoted tumor initiation and progression. Taken together, these findings highlight a previously unappreciated mechanism for activation of NF-κB signaling by OXCT1 and ketone body, and demonstrate that targeting OXCT1 can inhibit NSCLC tumorigenesis.